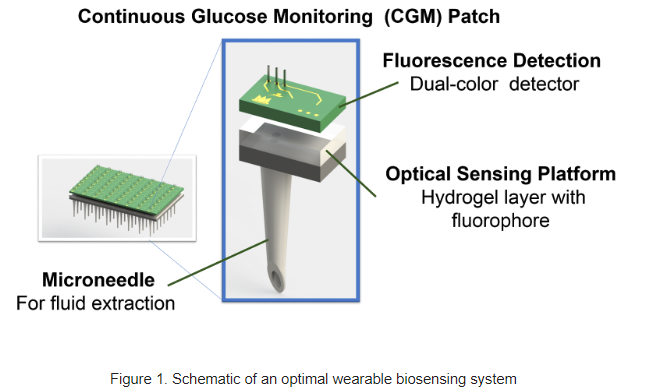
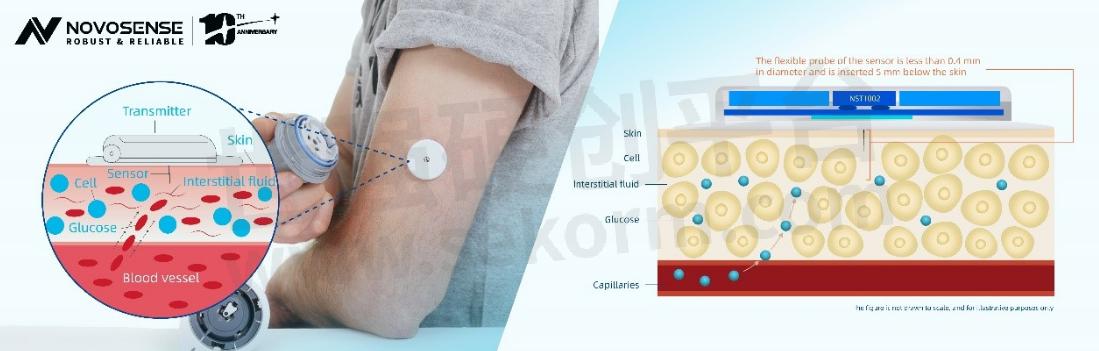
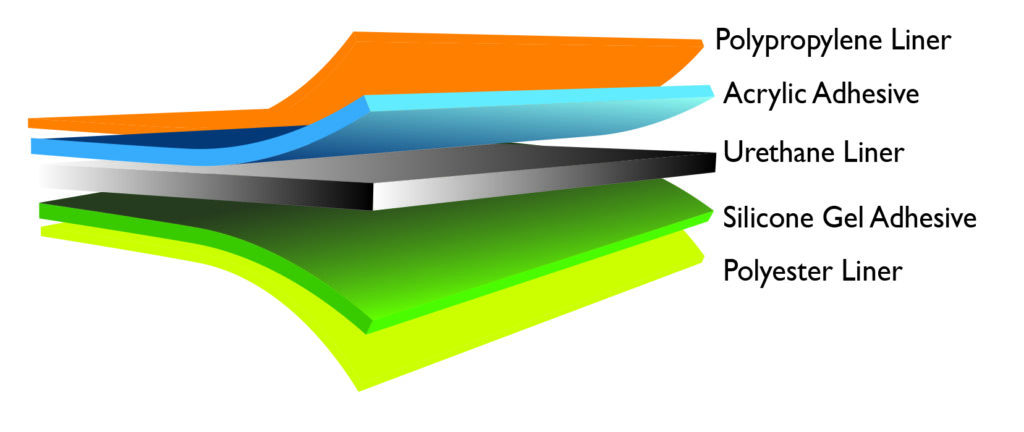
Engineering CGM Sensor Design: A Comprehensive Guide
The world of continuous glucose monitoring (CGM) systems has witnessed a significant shift from traditional blood glucose meter (BGM) technology. The advancements in electrode design, material selection, and sensor performance have paved the way for the development of more accurate and reliable CGM systems. In this article, we will delve into the principles of CGM systems, focusing on the core sensing technologies, outer membrane design, and key performance indicators.
Principles of CGM Systems
CGM systems are designed tomeasure glucose levels in the subcutaneous space via a sensor that includes wireless transmission of data to a receiving device, such as a smartphone. The system should include the following components:
- Glucose sensor: The sensor measures glucose levels in the interstitial fluid (ISF) through electrochemical reactions.
- Transmission module: The module transmits the glucose readings wirelessly to a receiving device.
- Receiving device: The device displays the glucose readings and allows users to track their glucose levels.
The core sensing technology in CGM systems involves electrochemical reactions that measure glucose levels in the ISF. The most common electrochemical sensing mechanisms include:
- Amperometric sensing: This method measures the current generated by the oxidation or reduction of glucose molecules.
- Enzymatic sensing: This method uses an enzyme to catalyze the reaction that measures glucose levels.
- Ion-sensitive field-effect transistor (ISFET) sensing: This method uses an ISFET to measure the pH change caused by glucose levels.
Outer Membrane Design
The outer membrane of the CGM sensor plays a crucial role in maintaining the integrity of the sensor and ensuring accurate glucose readings. The membrane should be designed to:
- Prevent contamination and interference from external factors.
- Allow for efficient glucose diffusion and transfer.
- Be biocompatible and non-toxic.
Key Performance Indicators
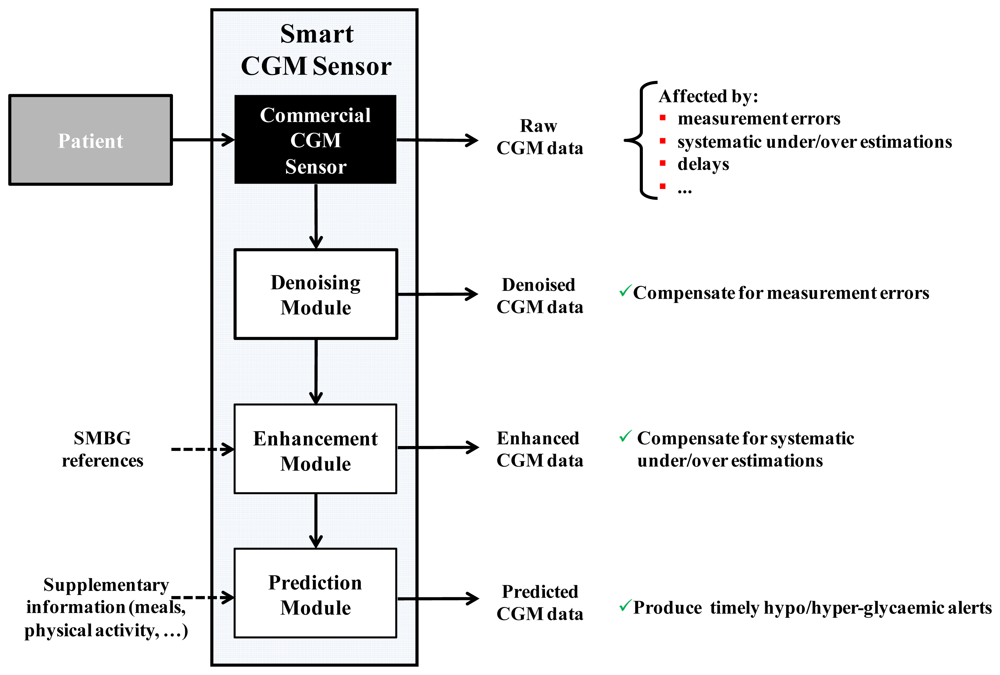
The key performance indicators (KPIs) for CGM systems include:
- Accuracy: The accuracy of the glucose readings in relation to the reference standard.
- Repeatability: The consistency of the glucose readings over time.
- Linearity: The ability of the sensor to accurately measure glucose levels across the entire range.
- Stability: The ability of the sensor to maintain accurate readings over time.
Design Considerations for CGM Systems
When designing CGM systems, engineers should consider the following factors:
- Size and weight: The device should be compact and lightweight for user comfort.
- Power consumption: The device should have a low power consumption to extend battery life.
- Wireless connectivity: The device should have a reliable and secure wireless connection to the receiving device.
- Biocompatibility: The device and its components should be biocompatible and non-toxic.
Conclusion
CGM systems have revolutionized the management of diabetes by providing accurate and reliable glucose readings without the need for fingersticks or scanning. The development of CGM systems requires careful consideration of the core sensing technologies, outer membrane design, and key performance indicators. By understanding these principles and design considerations, engineers can design CGM systems that meet the rising demand for ultra-compact and discreet devices that offer greater reliability, safety, and convenience for users with diabetes.
References:
Note: