The Mysterious World of Atoms
A lot can be said about the concept of "only want til atom." In reality, most atoms don't want to have 8 atoms, as this is just a simplification taught in school. Many atoms want more, and the actual number is 2, 8, 18, 32, 32, and so on. However, some special atoms don't want a full outer shell.
Understanding the Octet Rule
Atoms are the building blocks of matter, and they play a crucial role in forming chemical bonds. The octet rule is a fundamental concept in chemistry that explains how atoms tend to gain, lose, or share electrons to achieve a full outer shell, which consists of 8 electrons. This rule is based on the idea that atoms want to be in a stable state and reach a full outer shell to achieve this stability.
- Atomic Number:** The atomic number of an atom determines the number of electrons it has in its outer shell. Atoms with a low atomic number tend to lose electrons, while those with a high atomic number tend to gain electrons.
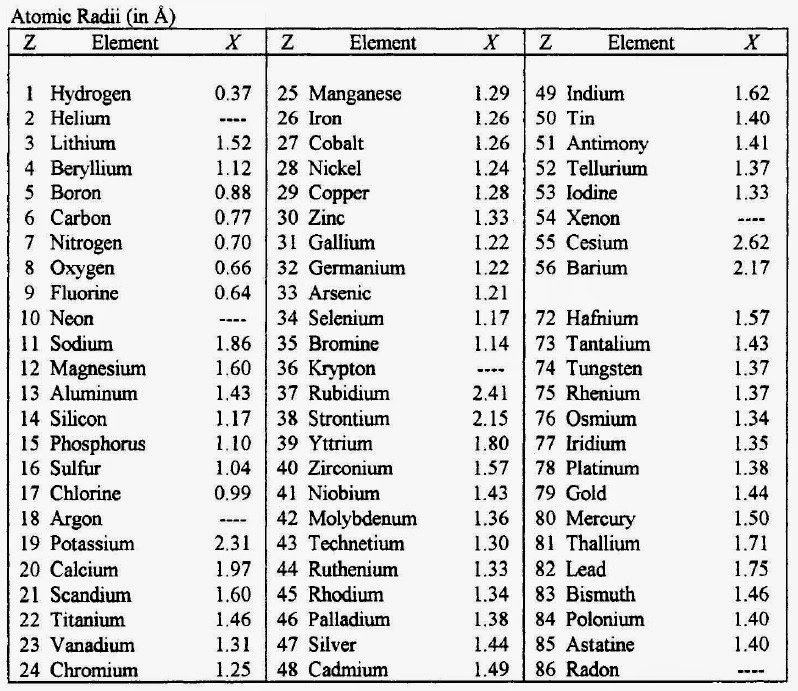
- Isotopes:** Atoms can have different numbers of neutrons, which affects their mass but not their chemical properties. The isotopes of an element have the same number of protons but differ in the number of neutrons.
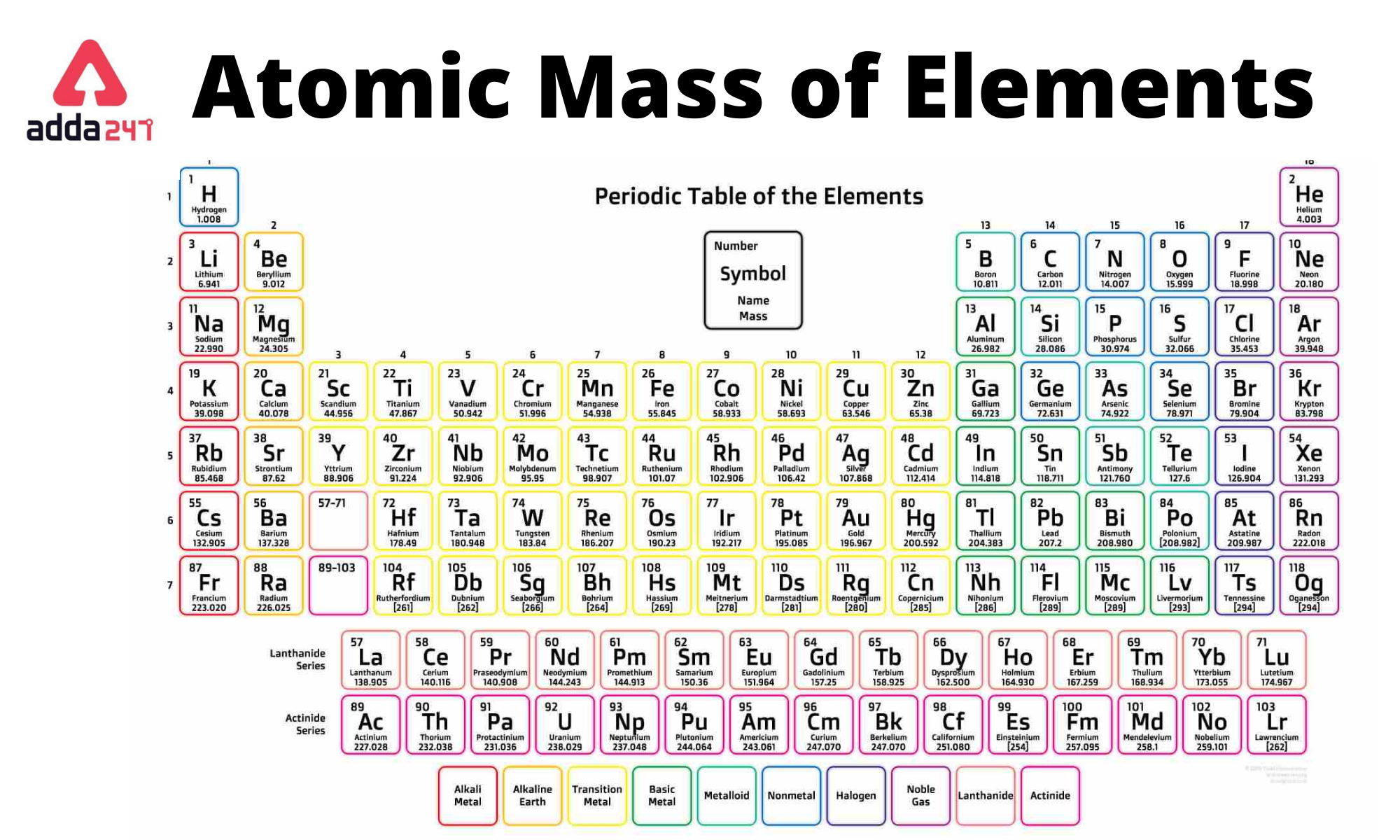
- The Periodic Table:** The periodic table organizes elements based on their atomic number and electron configuration. Elements with similar properties are grouped together in the periodic table.
Why Atoms Want a Full Outer Shell
Atoms want a full outer shell because it provides them with a stable configuration. When an atom has a full outer shell, it is less likely to react with other atoms, which reduces its reactivity and makes it more stable. This stability is due to the fact that a full outer shell has a lower energy state compared to an incomplete shell.
Take the example of oxygen, which has 8 protons and 8 electrons. Oxygen wants to have 8 valence electrons to achieve a full outer shell. However, if it gains 2 more electrons, it will have a charge of -2. This shows that oxygen wants to achieve a full outer shell to become stable and less reactive.
Exceptions to the Octet Rule
Not all atoms follow the octet rule, and some exceptions include:

- Hydrogen:** Hydrogen only needs 2 electrons to form a stable bond, which is why it doesn't follow the octet rule.
- Boron:** Boron has 3 valence electrons, which means it only needs 3 electrons to form a stable bond.
- Carbon:** Carbon has 4 valence electrons and can form stable bonds with 4 electrons in its outer shell.
Real-World Applications of Atoms
Atoms are the building blocks of everything around us, from the food we eat to the air we breathe. Understanding how atoms work is crucial for developing new technologies, such as:
- Nuclear Power:** Nuclear power plants use atoms to generate electricity.
- Materials Science:** Materials scientists study the properties of atoms to develop new materials with unique properties.
- Medicine:** Understanding how atoms interact with each other is crucial for developing new medicines and treating diseases.
In Conclusion
Atoms are the fundamental building blocks of matter, and understanding how they work is crucial for developing new technologies and improving our lives. While the octet rule provides a general framework for understanding how atoms interact with each other, there are exceptions and nuances that need to be considered. By studying atoms and their properties, we can unlock new understanding and develop new technologies that improve our world.
References
Bohr, N. (1913). The Structure of the Atom. In Nobel Lectures (pp. 123-144).
Heisenberg, W. (1927). The Uncertainty Principle. Zeitschrift für Physik, 43(3-4), 166-178.